Highlight
Investigating the unique properties of multiscale particles for pharmaceutical applications
Achievement/Results
NSF-funded researchers at Rutgers University are examining the synthesis, characterization, and function of novel multiscale particles for pharmaceutical applications. Associate Professor Nina Shapley joined the Department of Chemical and Biochemical Engineering and the Nanopharmaceutical IGERT program in September, 2008, after six years as an Assistant Professor in the Department of Chemical Engineering at Columbia University. At Columbia, Prof. Shapley established a dynamic, interdisciplinary research program that combines multiphase fluid mechanics, imaging, and design of biocompatible particles. In current work at Rutgers, she is concentrating on the aspects of the research related to pharmaceutical applications. She is currently advising two graduate students, Kristin Gilida and Kapil Deshpande, who are both supported by NSF IGERT fellowships, and four undergraduate students in multiscale micro- and nanoparticle research (see Figure 1). Half of the graduate and undergraduate advisees are female, thus supporting efforts of the IGERT program to enhance the diversity of the research environment.
Each of the IGERT fellows is pursuing a detailed fundamental study of multiscale particles:
1. Collaborative Research: Understanding protective encapsulation mechanisms through hybrid nanoarchitectures (Kristin Gilida)
A key aspect of the delivery of therapeutic biomolecules through routes that are alternatives to injection is the need to protect the active drug biomolecule from moisture, UV radiation and oxygen in the surrounding environment. There is great potential for encapsulation techniques to enhance the photo-oxidative and moisture stability of biological drug molecules, but the design and evaluation of protective polymer particles is currently at a very early stage of development. In this study, we aim to develop novel architectures for drug protectant materials. In addition, the encapsulation in a transparent polymer of a highly visible natural pigment (e.g. beta carotene) can be utilized to produce optically detectable tracer particles for assessing the fate and transport of encapsulated drugs in biological tissues. Next generation tracer particles can also carry contrast agents for standard noninvasive clinical imaging techniques. An important function of the encapsulating particle is to shield the pigment or contrast agent from degradation during the process of transport and detection.
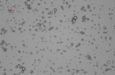
Our study explores new hybrid architectures, incorporating polymers, nanoparticles, and biological drug molecules or natural dyes, in order to elucidate the relationship between the spatial distribution of protectant molecules (e.g. UV absorbers and stabilizers) and stabilization efficiency. Initial experiments focus on the encapsulation and protection of beta carotene, which is both a drug molecule and natural pigment. A bright field image of core-shell microparticles containing encapsulated beta carotene is shown in Figure 2. Understanding the mechanisms that stabilize the encapsulated beta carotene is important for developing effective drug delivery vehicles tailored for the surrounding environment. Encapsulation of a natural pigment such as beta carotene can also be utilized to produce tracer particles for tracking the progress of encapsulated therapeutic materials in vivo. Since optical characterization is a key part of experiments with model nanoarchitectures, a transparent polymer matrix (PMMA) is initially being implemented. Biocompatible encapsulating polymers will be utilized in subsequent experiments.
2. Formation of multiscale biopolymer particle structures for improved nanoparticle handling (Kapil Deshpande)
A current challenge in pharmaceutical processing is the stabilization of suspensions containing nanoscale therapeutic particles so that particle agglomeration is avoided during manufacture of the dosage form. We hypothesize that the formation of ordered multiscale structures involving microparticles surrounded by therapeutic nanoparticles can lead to more precise nanoparticle handling with minimal uncontrolled agglomeration. Cohesive mechanisms include electrostatic attraction between the micro and nanoparticles and differential wetting of nanoparticles at an oil-water interface. The concept of hierarchical ordered particle structuring has been systematically developed for dry particle coating, and this work aims to extend the idea to aqueous systems. The combined, hierarchical structures suspended in an aqueous solution can then be ordered by oscillatory shear flow or electric fields, potentially exhibiting a more uniform spatial distribution of nanoparticles on the macroscopic length scale than can be obtained from uncontrolled suspensions of nanoparticles. The goal of this study is to enhance understanding of the mechanisms of hierarchical particle formation and particle size separation during processing of pharmaceutical suspensions. Specifically, the work will examine multiphase flow in complex geometries that mimic nozzles and coating devices in order to discover the mechanisms driving particle size separation phenomena during flow and resulting nanoparticle distributions. Utilization of abundant and biocompatible natural biopolymers, such as alginate and chitosan, with low energy cross-linking routes offers a unique opportunity for flexibility of dosage form design.
The specific aims of the project include: (1) Synthesizing biopolymer microbeads coated with complementary nanoparticles and characterizing morphology and stability during flow. (2) Understanding mechanisms of particle size separation in a bimodal suspension flowing through model coating and nozzle (contraction-expansion) flow geometries (see Figure 3). (3) Quantifying the macroscale uniformity of nanoparticles in the resulting model product. Improved insight into the multiphase transport aspects of active nanoparticle handling is essential for improving the efficacy and safety of nanoscale drug components.
Address Goals
Discovery: The development of multiscale polymer particles for pharmaceutical applications is a uniquely interdisciplinary endeavor that involves multiple aspects of micro- and nanoscale systems, including cross-linking processes in polymers, emulsification, phase inversion and precipitation, micro- and nanoencapsulation, the behavior of particles in a suspension flowing through a complex geometry, and the application of imaging techniques to evaluate particle spatial distribution and functional properties. Hence, the results of the study can transfer new concepts among several research areas, and are relevant to a host of applications.
The research from the first project described here can guide the development of multifunctional protective materials into future pharmaceutical technology in the areas of drug storage and delivery and tracer particles for use in vivo. The research is aimed toward aiding design of the next generation of suspension-based tablet coatings, spray-dried powders and thin films. In the second project, the fundamental research results can be implemented as long-term practical guidelines for the utilization of complementary biopolymer micro- and nanoparticle structures in novel pharmaceuticals, in order to improve efficiency of delivery and employ biocompatible and environmentally benign materials. One great advantage of the multiscale particles examined here is the ease of production, where detailed chemical synthesis is not required, and therefore the barrier to adoption is quite low. The results will be disseminated in journal publications and conference presentations.
Learning: This research investigation provides excellent training for graduate students Kristin Gilida and Kapil Deshpande in polymer micro- and nanoparticle synthesis and characterization, complex fluid rheology, imaging with electron microscopy, image analysis using Matlab and ImageJ software, literature review, and data interpretation. Since both projects involve collaborative work, graduate students are exposed to the research expertise of chemists and pharmaceutical scientists, as well as that of chemical engineers. In addition, the advisor and half of the graduate and undergraduate advisees are female, thus supporting efforts of the IGERT program to enhance the diversity of the research environment. Prof. Shapley is experienced in mentoring women in her laboratory after graduating two female Ph.D. students and advising two female M.S. students and nine female undergraduates at Columbia University. In summary, multiple aspects of the research program promote a stimulating and nurturing atmosphere leading to effective learning.