Highlight
Development and Characterization of Polymers for Biomedical Applications and Gene Therapies
Achievement/Results
NSF-funded researchers, Dr. Tim Long (Chemistry), Dr. Theresa Reineke (Chemistry), Webster Santos (Chemistry), Padma Rajagopalan (Chemical Engineering), and Rick Davis (Chemical Engineering) with MILES-IGERT trainees Rebecca Brown, Matt Green, John Layman, Mike Allen, Giovana Grandinetti, David Bryson, and Adam Larkin are designing and synthesizing polymers and peptide sequences for unique and varied biomedical applications.
Rebecca is working on development and characterization of charged polymers containing both positive and negative charges. These polymers have broad applications including formation of membranes to mimic those in biological systems, formation into fibers by a process called electrospinning, and use as antimicrobials.
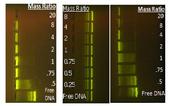
John, Matt and Mike are developing polymers that are specifically suited to gene therapy. In gene therapy, plasmid DNA encoded with therapeutic proteins is delivered to the cell nucleus, where protein undergoes several processes to express the therapy. Due to the negatively charged nature of DNA, a positively charged vector is needed to shield the charge as well as chaperone the DNA into the cell. This occurs through the generation of electrostatic complexes referred to as polyplexes. John, Matt, and Mike are conducting research with the aim of formulating systematic polymer structure-property relationships that will help elucidate polymer parameters that impact and optimize nucleic acid delivery.
John Layman has synthesized and characterized water-soluble macromolecules in both neutral and charged forms (linear and branched poly(2-N,N-dimethylaminoethyl methacrylate). John was also working to retrofit polymers with recognition molecules capable of honing in on cellular-specific surface proteins. Matt Green has synthesized and characterized a new potential nonviral vector for gene delivery (alkylated poly(1-vinylimidazole)) and evaluated the effect of charge density on formation of polyplexes. These researchers also are evaluating the cytotoxicity of the polymers and transfection efficiency. Mike Allen has successfully synthesized two monomers and two novel polymers that could lead to better gene delivery polymers. These polymers also are well suited for attaching drugs, antioxidants, or other compounds for additional biological purposes. Mike is currently developing additional highly functional monomers and will eventually evaluate the transfection efficiency and cytotoxicity of the polymers he develops.
Giovana Grandinetti and Dr. Theresa Reineke are studying how the mechanism of nuclear entry affects toxicity of cationic gene delivery polymers. Giovana has focused on elucidating the role that the mitochondrial permeability transition pore plays in polymer transfection. David Bryson and Professor Santos have synthesized and screened a library of branched peptides comprised of 4,096 unique peptide sequences for the purpose of selectively binding specific RNA sequences. In this research, branched peptide libraries were synthesized on a solid-phase support. All of the peptides were screened on the solid support for binding to HIV-1 TAR RNA, which is the region of the HIV RNA that is responsible for efficient viral replication. They found that several of the peptides were able to selectively bind to the 29 nucleotide RNA sequence over a mutant RNA sequence that varied by a single nucleotide. This group is currently analyzing where the peptides are binding to the target RNA sequence using radioactively labeled RNA sequences.
Adam Larkin, Padma Rajagopalan and Rick Davis are building biocompatible polymer films based on electrostatic interactions between the polymer molecules. These films are assembled under cellular conditions and are currently being characterized. The researchers are using models to predict the wet thickness of the films and have estimated them to be in the range of 1 to 10 nm each. These film thicknesses are suitable for use in understanding how different liver cells interact with each other as function of the separation distance provided by the polymer films.
Address Goals
Gene therapy is currently a very active field, primarily because of the potential for treatment of a variety of genetic diseases. Understanding the formation, size, charge, composition, and dissociation of polyplexes could lead to further understanding of the therapy delivery, which would provide information useful for designing better vectors, therapeutic drugs, or antioxidants. Understanding the mechanism for transfection presents other possibilities such as observing the transfection of cells using novel and classic antioxidative materials escorted by charged biomacromolecules. An understanding of the oxidative mechanism and its effect on cell structure and function, which are themselves biomacromolecules, would provide us with information useful for engineering antioxidative therapies that could be transfected into the cell via specifically engineered non-viral vectors. Transfection of radical scavenging molecules would allow restoration of the oxidant/anti-oxidant system in the cell. Synthesis and screening of peptides is important work because the number of proteins in the body that can currently be targeted by drugs is relatively limited. Thus, by being able to control protein production through binding of precursory RNA, a new and promising way of treating diseases could be developed.